Advertisements
Advertisements
प्रश्न
AB3 is an interhalogen T-shaped molecule. The number of lone pairs of electrons on A is ______. (Integer answer)
विकल्प
9
6
2
0
MCQ
रिक्त स्थान भरें
उत्तर
AB3 is an interhalogen T-shaped molecule. The number of lone pairs of electrons on A is 2.
Explanation:
AB3 represent sp3d hybridisation and it contains 3 bond pair and 2 lone pair.
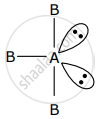
Number of lone pair = 2
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
