Advertisements
Advertisements
प्रश्न
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
उत्तर
Balanced equations for the purification of bauxite:
\[\ce{Al2O3.2H2O + 2NaOH -> 2NaAlO2 + 3H2O}\]
\[\ce{NaAlO2 + 2H2O -> NaOH + Al(OH)3 ↓}\]
\[\ce{2Al(OH)3 ->[heat][1000^\circ C]Al2O3 + 3H2O}\]
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Give the importance of the following for living beings:
Hydrogen
An ore on being heated in air forms sulphurous anhydride. Write the process used for the concentration of this ore.
`Cu (OH)2` ................................
................................
`ZnCO_3`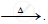 ..............................
..............................
`HgS + O_2`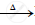 ..................
..................
Arrange the metals in (2) in the decreasing order of reactivity.
Complete the following flow chart and answer the questions below:
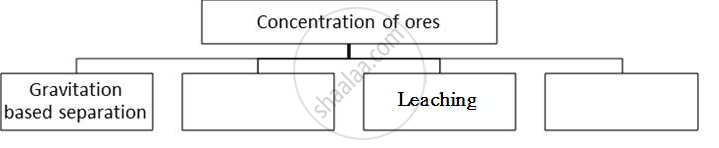
i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
Explain the hydraulic separation method with a neat labelled diagram.
Explain the following term:
Metallurgy
