Advertisements
Advertisements
प्रश्न
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
उत्तर
Balanced equations for the purification of bauxite:
\[\ce{Al2O3.2H2O + 2NaOH -> 2NaAlO2 + 3H2O}\]
\[\ce{NaAlO2 + 2H2O -> NaOH + Al(OH)3 ↓}\]
\[\ce{2Al(OH)3 ->[heat][1000^\circ C]Al2O3 + 3H2O}\]
APPEARS IN
संबंधित प्रश्न
Give the importance of the following for living beings:
Nitrogen
Give the importance of the following for living beings:
Carbon
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: carbonates
A student has been collecting silver coins and copper coins. One day she observed a black
coating on silver coins and a green coating on copper coins. Which chemical phenomenon is
responsible for these coatings? Write the names of black and green coatings.
`ZnCO_3`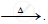 ..............................
..............................
`2Pb(NO3)_2`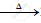 .........................
.........................
Which metal is used for:
lithographic plates for printing
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
Observe the figure and name and explain in brief the following method:

Limestone is used as a flux in the extraction of ______.
