Advertisements
Advertisements
प्रश्न
An electronic displacement in a covalent bond is represented by the following notation.
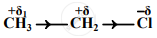
A. Identify the effect
B. Is the displacement of electrons in a covalent bond temporary or permanent.
उत्तर
- The electronic displacement represented above is the inductive effect (–I effect).
- The inductive effect is a permanent electronic effect as it depends on the electronegativity of the atoms. In the given example, the displacement of electrons is permanent as Cl is more electronegative than C.
APPEARS IN
संबंधित प्रश्न
Write IUPAC names of the following.

Write the IUPAC name of the following.
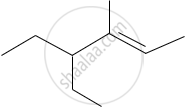
Write the IUPAC name of the following.

Observe the following structures and answer the questions given below.
- \[\ce{CH3 - CH2 - CH2 - CHO}\]
\[\begin{array}{cc}\ce{CH3 - CH - CHO}\\
|\phantom{...}\\\ce{CH3}\end{array}\]
a. What is the relation between (i) and (ii)?
b. Write IUPAC name of (ii).
c. Draw the functional group isomer of (i).
The general formula for alkadiene is ______.
In the hydrocarbon
![]() the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
The IUPAC name of the compound\[\begin{array}{cc}\ce{CH3-CH=C-CH2-CH3}\\
|\phantom{..}\\\phantom{...............}\ce{CH2 - CH2 - CH3}\end{array}\] is
The structure of isobutyl group in an organic compound is
Give the IUPAC names of the following compound.
\[\ce{CH2 = CH - CH = CH2}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - C ≡ C - CH - CH3}\\
\phantom{........}|\\\phantom{.........}\ce{Cl}
\end{array}\]
Give the IUPAC names of the following compound.
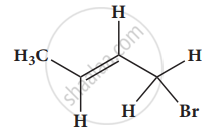
Give the IUPAC names of the following compound.
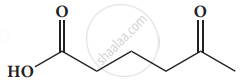
Give the IUPAC names of the following compound.

Give the IUPAC names of the following compound.
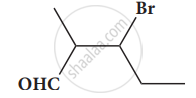
Give the structure for the following compound.
3 – ethyl – 2 methyl – 1 – pentene
Give the structure for the following compound.
3 - Chlorobutanal
Give the structure for the following compound.
3 - Chlorobutanol
Give the structure for the following compound.
Octane - 1,3- diene
Give the structure for the following compound.
3-Chlorobut - 1 - ene
Give the structure for the following compound.
acetaldehyde
Which among the following carbocation is most reactive?
lUP AC name of
\[\begin{array}{cc}
\phantom{}\ce{C2H5}\phantom{.}\ce{Cl}\phantom{....}\ce{CH3}\phantom{......}\\
\phantom{}|\phantom{.....}|\phantom{......}|\phantom{........}\\
\ce{H3C - CH2 - CH - CH - CH - CH2 - CH2 - CH3}
\end{array}\]
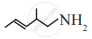
What is the IUPAC name of the following compound?
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH2 - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{.....}|\phantom{.............}|\phantom{...............}\\
\phantom{}\ce{CH3}\phantom{.}\ce{CH3}\phantom{..........}\ce{C2H5}\phantom{...........}
\end{array}\]
What is the IUPAC name of the following compound?
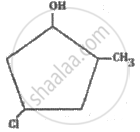
The IUPAC name of the following compound is:
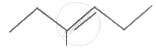
The IUPAC name of the following compound is:
CH2 = C = CH − CH3
The IUPAC name of the following compound is: