Advertisements
Advertisements
प्रश्न
An electronic displacement in a covalent bond is represented by the following notation.
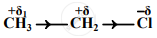
A. Identify the effect
B. Is the displacement of electrons in a covalent bond temporary or permanent.
उत्तर
- The electronic displacement represented above is the inductive effect (–I effect).
- The inductive effect is a permanent electronic effect as it depends on the electronegativity of the atoms. In the given example, the displacement of electrons is permanent as Cl is more electronegative than C.
APPEARS IN
संबंधित प्रश्न
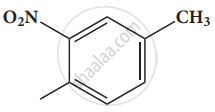
Write the IUPAC name of the following.
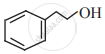
Write the IUPAC name of the following.

Phytane is a naturally occurring alkane produced by the alga spirogyra and is a constituent of petroleum. The IUPAC name for phytane is 2,6,10,14-tetramethylhexadecane. Write a zig-zag formula for phytane. How many primary, secondary, tertiary, and quaternary carbons are present in this molecule?
| A covalent bond in tert-butyl bromide breaks in a suitable polar solvent to give ions. |
- Name the anion produced by this breaking of a covalent bond.
- Indicate the type of bond breaking in this case.
- Comment on the geometry of the cation formed by such bond cleavage.
The general formula for alkadiene is ______.
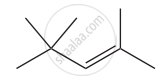
The IUPAC name of the compound is
Which one of the following names does not fit a real name?
IUPAC name of \[\begin{array}{cc}
\phantom{....}\ce{H}\phantom{...}\ce{C4H9}\\
|\phantom{....}|\\\ce{CH3 - C - C - CH3}\\
|\phantom{....}|\\\phantom{.....}\ce{C2H5}\phantom{.}\ce{CH3}\phantom{...}\end{array}\] is
The IUPAC name of \[\begin{array}{cc}
\phantom{.}\ce{CH3}\\|\phantom{..}\\
\ce{H3C - C - CH = C(CH3)2}\\
|\phantom{..}\\\phantom{..}\ce{CH3}
\end{array}\] is
The structure of isobutyl group in an organic compound is
Give the IUPAC names of the following compound.
\[\ce{(CH3)2 CH–CH2 –CH(CH3 )–CH(CH3)2}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
\phantom{...}|\phantom{......}|\phantom{.....}\\
\ce{CH3}\phantom{...}\ce{Br}\phantom{.}
\end{array}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - C ≡ C - CH - CH3}\\
\phantom{........}|\\\phantom{.........}\ce{Cl}
\end{array}\]
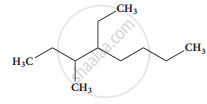
Give the IUPAC names of the following compound.
Give the IUPAC names of the following compound.
Give the structure for the following compound.
3 – ethyl – 2 methyl – 1 – pentene
Give the structure for the following compound.
1,3- Dimethylcyclohexane
Give the structure for the following compound.
3 - methylbutan - 2 - ol
As per IUPAC nomenclature, the name of the complex Na3[AlF6] is ____________.
In the reaction, \[\ce{Anisole + {'A'} ->[Anhydrous][AlCl3] 4-Methoxyacetophenone}\]
'A' is ____________.
IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3 - C - CH2CH3}\\
|\phantom{....}\\
\ce{OH}\phantom{..}
\end{array}\]
IUPAC name of
\[\begin{array}{cc}
\ce{C2H5 - CH2 - C(Cl) - C2H5}\\
\phantom{...}|\\
\phantom{......}\ce{C2H5}
\end{array}\]
The IUPAC name of the following compound is:
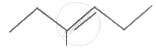
The IUPAC name of the following compound is:
CH2 = C = CH − CH3
The correct structure of 2,6-Dimethyl-dec-4-ene is ______
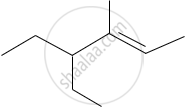
The IUPAC name of the following compound is:
Consider the following reactions.
\[\ce{C6H5N^+_2Cl- ->[HBF4] A ->[NaNO2/Cu] B ->[Sn/HCl] C}\]
Identify the compound formed C in the above reaction.
