Advertisements
Advertisements
प्रश्न
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
उत्तर
The difference in the acidic character of phenols and alcohol is due to the difference in reactivity of these compounds towards the ionization of the O–H bond. This can be explained as follows:
i. Ionization of alcohols is represented by the following equilibrium
\[\ce{\underset{\text{Alcohol}}{R - OH} + H2O ⇌ \underset{\text{Alkoxide}}{R - O-} + H3O+}\]
The electron-donating inductive effect (+I effect) of the alkyl group destabilizes the alkoxide ion (the conjugate base of alcohol). As a result, alcohol does not ionize much in the water, and behaves like a neutral compound in an aqueous medium.
ii. Ionization of phenol is represented by the following equilibrium
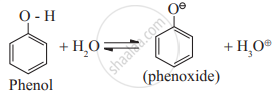
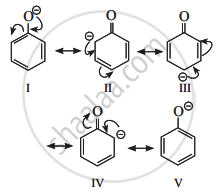
Phenoxide ion, the conjugate base of phenol, is resonance stabilized by delocalization of the negative charge. Therefore, phenol ionizes in an aqueous medium to a moderate extent. Thus phenols are more acidic than ethyl alcohol.
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Answer in brief.
Give a simple chemical test to distinguish between ethanol and ethyl bromide.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Write IUPAC name of crotonyl alcohol.
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
How will you bring about the following conversions?
acetone to 2-methylpropan-2-ol
Write the reactions for the preparation of carbolic acid from aniline.
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Phenol is obtained from cumene ____________.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
Phenol is ____________.
Which of following elements does not react with hot concentrated sulphuric acid?
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
Identify 'A' and 'B' in the following series of reactions.
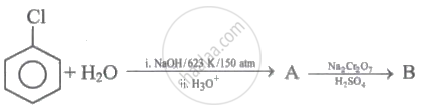
In Dow process, chlorobenzene is converted into phenol by ____________.
Which of the following alcohols has tertiary allylic carbon?
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which isomer of C4H10O is optically active?
Which among the following compounds is used to prepare solid fuel using calcium acetate?
Which of the following alcohols is least soluble in water?
Which among the following is allylic secondary alcohol?
Identify the alcohol that reacts immediately with Lucas reagent?
Which of the following alcohols is NOT prepared by reduction of carbonyl compounds?
Which among the following phenolic compound is most acidic in nature?

Product (B) in this reaction is:
Arrange the following compounds in an increasing order of their solubility in water:
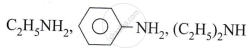
Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
