Advertisements
Advertisements
प्रश्न
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
उत्तर
Ethyl alcohol gives hydrogen on reaction with sodium metal. On oxidation with PCC ethanol will form acetaldehyde (C2H4O).
\[\ce{\underset{\text{Ethyl alcohol}}{2CH3 - CH2 - OH} ->[2Na] \underset{\text{Sodium ethoxide}}{2CH3 - CH2 - O- Na+} + {H_{2(g)}} ^}\]
\[\ce{\underset{\text{Ethyl alcohol}}{CH3 - CH2 - OH} ->[{[O]}][PCC] \underset{\ce{Acetaldehyde(C2H4O)}}{CH3 - CHO} + H2O}\]
संबंधित प्रश्न
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Reaction between hot conc. HI and anisole gives ______________
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Which of the following compounds is obtained, when phenol react with bromine water?
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
The secondary alcohol is ____________.
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
Identify 'A' and 'B' in the following series of reactions.
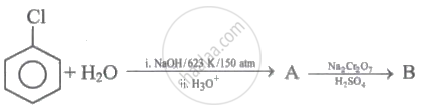
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
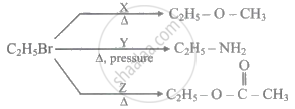
Identify reagents X, Y and Z.
Which among the following compounds is used to prepare solid fuel using calcium acetate?
Which of the following alcohols is least soluble in water?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the role of PDC from the following.
The compound which reacts fastest with Lucas reagent at room temperature is ______.
Which among the following phenolic compound is most acidic in nature?

Product (B) in this reaction is:
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
Which among the following is not the method of preparation of phenol?
The major product obtained in the following reaction is

What is the action of nitrous acid on aniline?
Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Explain: Phenols are acid while alcohol is neutral.
