Advertisements
Advertisements
प्रश्न
Explain the fact that in alkyl aryl ethers, the alkoxy group is ring activating and ortho/para directing towards electrophilic aromatic substitution.
उत्तर
- Resonance structures of alkyl aryl ethers are as follows:
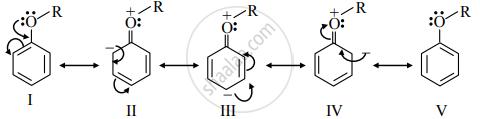
- The +R effect of −OR the group results in increased electron density at the para- and two ortho positions.
Hence, the alkoxy group in aromatic ether is a ring activating and ortho/para-directing group towards electrophilic aromatic substitution.
संबंधित प्रश्न
Choose the correct option.
Ethers react with cold and concentrated H2SO4 to form ________.
What is the action of atmospheric oxygen on ethers?
Write chemical equation of acetyl chloride with ethanol
Write the reaction between ethanol and acetic anhydride.
What is the action of following on diethyl ether?
dil. H2SO4
Which of the following compounds does not contain \[\begin{array}{cc}\backslash\phantom{......}\\\ce{C = O}\\
/\phantom{......}\\\end{array}\] group?
A mixed ether on hydrolysis gives ____________.
\[\ce{(CH3)2CH - O - CH3 ->[Cold HI] X + Y}\]
\[\ce{X ->[K2Cr2O7][dil. H2SO4] Z}\]
\[\ce{Y ->[NaOH(aq)][\Delta] CH3OH}\]
Identify X, Y and Z.
To prepare ethanol from methyl magnesium bromide, the other reagent required is ____________.
When methoxy benzene reacts with HI at room temperature the products formed are ______.
What are the products of the following reaction?
\[\ce{(CH3)3C - O - CH3 + HI ->[cold] ?}\]
Ether is obtained from ethyl alcohol in presence of H2SO4 at _______.
The major product of the reaction between tert-butyl chloride and sodium ethoxide is ______.
The compound which is not formed when a mixture of n-butyl bromide and ethyl bromide treated with sodium metal in the presence of dry ether is ______.
Isopropyl methyl ether when treated with cold hydrogen iodide gives ______.
Tert-butyl methyl ether on treatment with hydrogen iodide in cold gives ______.
Formation of diethyl ether from ethanol is based oh a ______.
Ethers when dissolved in cold concentrated sulphuric acid forms ______.
Why ethers possess a small net dipole moment?
Explain Williamson's synthesis.
Write preparation of diethyl ether.
