Advertisements
Advertisements
प्रश्न
Answer the following.
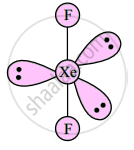
Draw structures of XeF2
उत्तर
Linear
APPEARS IN
संबंधित प्रश्न
Answer the following.
Name two gases which deplete ozone layer.
Answer the following.
Give one example showing reducing property of ozone.
What happens when lead sulfide reacts with ozone \[\ce{O3}\]?
Answer the following.
What happens when nitric oxide reacts with ozone.
Discuss the structure of sulfur dioxide.
Answer the following.
Draw structures of XeO3
Answer the following.
Draw structures of XeOF4
What are interhalogen compounds?
Answer the following.
How are xenon fluorides XeF2, XeF4 and XeF6 obtained? Give suitable reactions.
Answer the following.
How are XeO3 and XeOF4 prepared?
O2 molecule is ______.
The number of covalent bonds present in sulfuric acid:
Complete the following reaction:
\[\ce{SO_{2(g)} + Cl_{2(g)} ->[Charcoal]}\] ?
Identify the INCORRECT match.
Which among the following oxides of nitrogen is called nitrogen sesquioxide?
In ozone molecule the formal charge on the central oxygen atom is ______.
Find the CORRECT statement.
\[\ce{I2_{(g)} + H2S_{(g)} -> 2HI_{(g)} + S_{(s)}}\]
Which of the following statements is INCORRECT?
Mark the oxide which is amphoteric in character.
What will be the formula of an oxide of iodine (atomic mass = 127) which contains 25.4 g of iodine and 8g of oxygen?
Write balanced equation of a reaction in which ozone reduces hydrogen peroxide.
Write any two characteristics of interhalogen compounds.
Write the reaction of the following with concentrated H2SO4:
NaCl
Write the reaction of the following with concentrated H2SO4:
KNO3
What are the basic oxides? Explain with an example.
Electrolytic method of preparation of dioxygen.
Industrial method of preparation of dioxygen.
