Advertisements
Advertisements
प्रश्न
Answer the following question using the data given below:
- Assertion: The nature of bond in HF molecule is ionic
- Reason: The electronegativity difference between H and F is 1.9.
विकल्प
A and R are correct, R explains the A
A is correct, R is wrong
A is wrong, R is correct
A and R are correct, R doesn’t explain A
उत्तर
A and R are correct, R explains the A.
APPEARS IN
संबंधित प्रश्न
Rewrite the following sentences by using the correct symbol > (greater than) or < (less than) in the blanks given
The electronegativity of iodine is ___________ that of Chlorine
Define the term Electronegativity.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 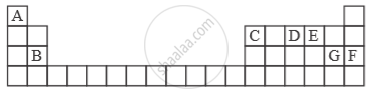
Which element forms electrovalent compound with G?
In terms of electronic configuration, what the elements of a given period and groups have in common?
Fill in the blank
Most electronegative elements belong to ______ group.
Explain
Electronegativity of Cl is higher than S.
Among the elements given below, the element with the least electronegativity is:
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The electronegativity:
Electronegativity across the period ______.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last). Answer the following question in relation to the given group of elements:
Which element would be expected to have the highest electronegativity?
