Advertisements
Advertisements
प्रश्न
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 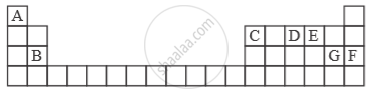
Which element forms electrovalent compound with G?
उत्तर
Electrovalent compound formed with ‘G’ is with ‘A’.
APPEARS IN
संबंधित प्रश्न
State the character of the oxide of period 3 ?
Among the elements of the second period, Li to Ne, pick out the element with the highest electro negativity
Select the correct answer
Which of the following element has highest electronegativity?
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Electro negativity increases going down a group.
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (decreasing electronegativity)
The element below sodium in the same group would be expected to have a ______ (lower/higher) electro-negativity than sodium and the element above chlorine would be expected to have a ______ (lower/higher) ionization potential than chlorine.
Answer the following question using the data given below:
- Assertion: The nature of bond in HF molecule is ionic
- Reason: The electronegativity difference between H and F is 1.9.
State the term for the following:
The tendency of an atom to pull a shared pair of electrons towards itself in a compound.
Identify the following:
The most electronegative element of Period 3.
