Advertisements
Advertisements
प्रश्न
The element below sodium in the same group would be expected to have a ______ (lower/higher) electro-negativity than sodium and the element above chlorine would be expected to have a ______ (lower/higher) ionization potential than chlorine.
उत्तर
The element below sodium in the same group would be expected to have a lower electro-negativity than sodium, and the element above chlorine would be expected to have a higher ionisation potential than chlorine.
APPEARS IN
संबंधित प्रश्न
Rewrite the following sentences by using the correct symbol > (greater than) or < (less than) in the blanks given
The electronegativity of iodine is ___________ that of Chlorine
Define the term Electronegativity.
Name the periodic property which relates to the tendency of an atom to attract the shared pair of electron.
This question refers to the elements of the periodic table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these are most electronegative element?
Arrange the elements of group 17 and group 1 according to the given conditions.
Decreasing electro negativity.
The tendency of an atom to attract electrons itself when combined in a covalent compound.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 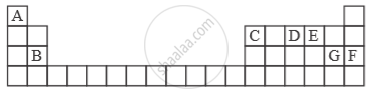
Which element forms electrovalent compound with G?
Among the elements given below, the element with the least electronegativity is:
Arrange the following as per the instruction given in the bracket
F,Cl,Br (Increasing electronegativity)
Electronegativity across the period ______.
