Advertisements
Advertisements
Question
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 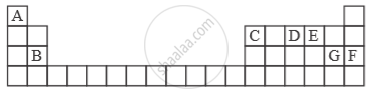
Which element forms electrovalent compound with G?
Solution
Electrovalent compound formed with ‘G’ is with ‘A’.
APPEARS IN
RELATED QUESTIONS
Name the periodic property which relates to the tendency of an atom to attract the shared pair of electron.
This question refers to the elements of the periodic table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these are most electronegative element?
Explain
K is more reactive than Li.
The electro negativities (according to Pauling) of the elements in period 3 of the periodic table are as follows.
The elements are arranged in alphabetical order:
Al Cl Mg Na P S Si
1.5 3.0 1.2 0.9 2.1 2.5 1.8
(i) Arrange the elements in the order in which they occur in the periodic table from left to right.
(The group 1 elements first, followed by the group 2 element and so on, up to group 7)
(ii) Choose the word or phrase from the brackets which correctly completes each of the following statements:
(a) The element below sodium in the same group would be expected to have a ______ (lower/higher) electro negativity than sodium and the element above chlorine which would be expected to have a ______ (lower / higher) ionization potential than chlorine.
(b) On moving from left to right in a given period, the number of shells ______ (remains the same / decreases/ increases).
(c) On moving down a group, the number of valence electrons ______ (remains the same / increases/ decreases).
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Electro negativity increases going down a group.
Among the elements given below, the element with the least electronegativity is:
If the electronegativity difference between two bonded atoms in a molecule is greater than 1.7, the nature of bonding is ______
Identify the following:
The most electronegative element of Period 3.
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (decreasing electronegativity)
Electronegativity across the period ______.
