Advertisements
Advertisements
Question
Among the elements given below, the element with the least electronegativity is:
Options
Lithium
Boron
Carbon
Fluorine
Solution
Lithium
Explanation:
Electronegativity increases from left to right. Lithium is present on the left side of the periodic table; hence, it will be the least electronegative element.
APPEARS IN
RELATED QUESTIONS
Name the elements with highest and lowest electronegativity ?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 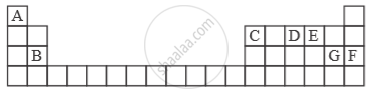
Which element forms electrovalent compound with G?
Fill in the blank
Most electronegative elements belong to ______ group.
Explain
K is more reactive than Li.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The electronegativity:
If the electronegativity difference between two bonded atoms in a molecule is greater than 1.7, the nature of bonding is ______
State the term for the following:
The tendency of an atom to pull a shared pair of electrons towards itself in a compound.
Identify the following:
The most electronegative element of Period 3.
Electronegativity across the period ______.
