Advertisements
Advertisements
प्रश्न
Answer the following question.
What is absolute zero?
उत्तर

Graph of pressure versus temperature (in °C) at constant volume.
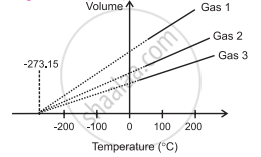
Graph of volume versus temperature (in °C) at constant pressure.
- When the graph of pressure (P) against temperature T (°C) at constant volume for three ideal gases A, B, and C is plotted, in each case, P-T graph is a straight line indicating direct proportion between them. The slopes of these graphs are different.
- The individual straight lines intersect the pressure axis at different values of pressure at 0 °C, but each line intersects the temperature axis at the same point, i.e., at absolute temperature (-273.15 °C).
- Similarly, graph at constant pressure for three different ideal gases A, B and C extrapolate to the same temperature intercept -273.15 °C i.e., absolute zero temperature.
- It is seen that all the lines for different gases cut the temperature axis at the same point at - 273.15 °C.
- This point is termed as the absolute zero of temperature.
- It is not possible to attain a temperature lower than this value. Even to achieve absolute zero temperature is not possible in practice.
APPEARS IN
संबंधित प्रश्न
What are the different scales of temperature? What is the relation between them?
Solve the following problem.
The volume of a gas varied linearly with absolute temperature if its pressure is held constant. Suppose the gas does not liquefy even at very low temperatures, at what temperature the volume of the gas will be ideally zero?
Solve the following problem.
A gas at 900 °C is cooled until both its pressure and volume are halved. Calculate its final temperature.
Solve the following problem.
An aluminium rod and iron rod show 1.5 m difference in their lengths when heated at all temperature. What are their lengths at 0 °C if coefficient of linear expansion for aluminium is 24.5 × 10–6/°C and for iron is 11.9 × 10–6/°C?
In a diesel engine, the cylinder compresses air from S.T.P. to about `1/14` the original volume and a pressure of 40 atmospheres. The temperature of compressed air is ______
The temperature of a body is 37°C on a Celsius scale, what is its temperature on the Kelvin scale?
Two moles of an ideal diatomic gas occupies a volume V at 27° C. The gas expands adiabatically to a volume 2V. Calculate the final temperature of the gas is nearly ____________.
One mole of a gas at pressure 2 Pa and temperature 37° C is heated till both pressure and volume are tripled. What is the temperature of the gas?
1 litre of an ideal gas at 27 °C is heated at a constant pressure to 127 °C. The final volume is approximately ______.
On a fast moving train, a container is placed enclosing some gas at 300 K, while the train is in motion, the temperature of the gas ______.
