Advertisements
Advertisements
प्रश्न
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (decreasing electronegativity)
उत्तर
Electron Negativity in a group decreases from top to bottom.
All these elements belong to group I.
Li > Na > K > Rb > Cs
संबंधित प्रश्न
Name the elements with highest and lowest electronegativity ?
State the character of the oxide of period 3 ?
This question refers to the elements of the periodic table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these are most electronegative element?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 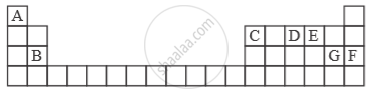
Which element forms electrovalent compound with G?
Fill in the blank
Most electronegative elements belong to ______ group.
Explain
K is more reactive than Li.
Explain
Electronegativity of Cl is higher than S.
Select the correct answer
Which of the following element has highest electronegativity?
The electro negativities (according to Pauling) of the elements in period 3 of the periodic table are as follows.
The elements are arranged in alphabetical order:
Al Cl Mg Na P S Si
1.5 3.0 1.2 0.9 2.1 2.5 1.8
(i) Arrange the elements in the order in which they occur in the periodic table from left to right.
(The group 1 elements first, followed by the group 2 element and so on, up to group 7)
(ii) Choose the word or phrase from the brackets which correctly completes each of the following statements:
(a) The element below sodium in the same group would be expected to have a ______ (lower/higher) electro negativity than sodium and the element above chlorine which would be expected to have a ______ (lower / higher) ionization potential than chlorine.
(b) On moving from left to right in a given period, the number of shells ______ (remains the same / decreases/ increases).
(c) On moving down a group, the number of valence electrons ______ (remains the same / increases/ decreases).
The electronegativities (according to Pauling) of the elements in period 3 of the periodic table are as follows with elements arranged in alphabetical order:
| Al | Cl | Mg | Na | P | S | Si |
| 1.5 | 3.0 | 1.2 | 0.9 | 2.1 | 2.5 | 1.8 |
Arrange the elements in the order in which they occur in the periodic table from left to right.
(The group 1 element first, followed by the group 2 element and so on, up to group 7).
