Advertisements
Advertisements
प्रश्न
Arrange the following as per the instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing order of metallic character).
उत्तर
Li < Na < K < Rb < Cs
APPEARS IN
संबंधित प्रश्न
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
Nature of oxides of the elements ?
How does the chemical reactivity of
alkali metals vary?
Choose the correct answer.
An alkaline earth metal
(i) Lead
(ii) potassium
(iii) calcium
(iv) Copper
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 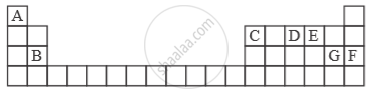
Which non-metallic element has the valency of 2?
In the third period, which is the most metallic and most non-metallic element?
Select the correct answer
Which of the following electronic structure is of a metal?
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The element at the bottom of a group would be expected to show ______ metallic character than the element at the top.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last). Answer the following question in relation to the given group of elements:
Which element has the most metallic character?
The metal which is hard and has a high melting point and used in filaments of electrical bulbs is ______
