Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 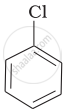 |
| (b) | 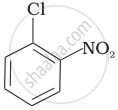 |
| (c) | 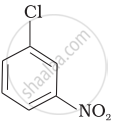 |
विकल्प
(a) < (b) < (c)
(c) < (b) < (a)
(a) < (c) < (b)
(c) < (a) < (b)
उत्तर
(a) < (c) < (b)
Explanation:
The presence of an electron-withdrawing group (−NO2) at the ortho and para position facilitates nucleophilic substitution. The effect of the presence of the electron-withdrawing group is very less at the meta position.
APPEARS IN
संबंधित प्रश्न
What happens when \[\ce{CH3 - Br}\] is treated with KCN?
Chlorobenzene is extremely less reactive towards a nucleophilic substitution reaction. Give two reasons for the same.
How the following conversion can be carried out?
Chlorobenzene to p-nitrophenol
Give reasons:
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride.
Write the product formed on reaction of D-glucose with Br2 water.
Assertion: Presence of a nitro group at ortho or para position increases the reactivity of haloarenes towards nucleophilic substitution.
Reason: Nitro group, being an electron-withdrawing group decreases the electron density over the benzene ring.
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 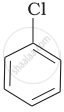 |
| (b) | 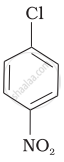 |
| (c) | 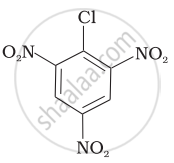 |
Haloarenes are less reactive than haloalkanes and haloalkenes. Explain.
Allyl chloride is hydrolysed more readily than n-propyl chloride. Why?
\[\ce{C6H12O6 ->[(Zymase)] A ->[NaOH][\Delta] B + CHI3}\]
The number of carbon atoms present in the product B is:
