Advertisements
Advertisements
प्रश्न
Arrange the following elements in order of their decreasing metallic character.
Na, Si, Cl, Mg, Al
विकल्प
Cl > Si > Al > Mg > Na
Na > Mg > Al > Si > Cl
Na > Al > Mg > Cl > Si
Al > Na > Si > Ca > Mg
उत्तर
Na > Mg > Al > Si > Cl
APPEARS IN
संबंधित प्रश्न
Give the trend in metallic character:
down the group top to bottom.
Name the periodic property which relates to the character of element which loses one or more electrons when supplied with energy.
Explain the following:
Metallic character of elements decreases from left to right in a period while it increases in moving down a group.
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
Nature of oxides of the elements ?
Choose the correct answer.
An alkaline earth metal
(i) Lead
(ii) potassium
(iii) calcium
(iv) Copper
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.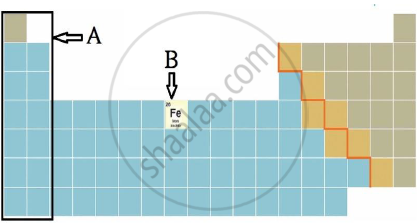
Mg2+, Cu2+, Na1+, H1+ (In the order of preferential discharge at the cathode)
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 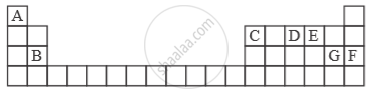
Which non-metallic element has the valency of 2?
Among the elements of the second period, Li to Ne, pick out the element that is the most reactive metal
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most metallic element be found at A,B,C or D ?
In the third period, which is the most metallic and most non-metallic element?
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The element at the bottom of a group would be expected to show ______ metallic character than the element at the top.
The tendency of an element to form cation is the _______ character of that element.
Electropositivity means _______.
On moving down the group metallic character increases.
Which of the following set of elements is written in order of their increasing metallic character?
Three elements B, Si and Ge are
Arrange:
Be, Li, C, B, N, O, F (in increasing metallic character).
