Advertisements
Advertisements
प्रश्न
Explain the following:
Metallic character of elements decreases from left to right in a period while it increases in moving down a group.
उत्तर
On moving across a period, nuclear pull increases because of the increase in atomic number, and thus, the atomic size decreases. Hence, elements cannot lose electrons easily. Hence, Group 17 elements are strong non-metals, while Group 1 elements are strong metals. Down a group, the atomic size increases and the nuclear charge also increases. The effect of an increased atomic size is greater as compared to the increased nuclear charge. Therefore, metallic nature increases as one moves down a group, i.e. they can lose electrons easily.
संबंधित प्रश्न
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Give the trend in metallic character:
(i) across the period left to right
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 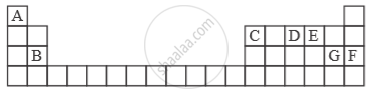
Which non-metallic element has the valency of 2?
Among the elements of the second period, Li to Ne, pick out the element that is the most reactive non-metal
Explain
Larger the atomic size, more metallic is the element.
Match the atomic number with the following:
A metal of valency one.
The tendency of an element to form anion is the ______ character of that element.
Electronegativity is the metallic property of an element.
Three elements B, Si and Ge are
