Advertisements
Advertisements
प्रश्न
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
उत्तर
On moving across a period, nuclear pull increases because of the increase in atomic number, and thus, the atomic size decreases. Hence, elements cannot lose electrons easily. Hence, Group 17 elements are strong non-metals, while Group 1 elements are strong metals.
संबंधित प्रश्न
Arrange the following as per the instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing order of metallic character).
Answer the following in respect of element `31/15 P `
What is its valency?
Write scientific reason.
Metallic character goes on decreasing while going from left to right in a period.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 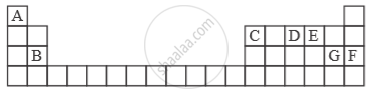
Which non-metallic element has the valency of 2?
State whether the following statement is true or false
All members of zero group are non metals.
In the third period, which is the most metallic and most non-metallic element?
Electropositivity means _______.
3, 1, 2 electrons are in valence shells of X, Y, Z elements. From this information, state the group in which they belong and write their valencies.
The lightest liquid metal is ______
Identify the elements with the following property and arrange them in increasing order of their reactivity
- An element which is a soft and reactive metal
- The metal which is an important constituent of limestone
- The metal which exists in liquid state at room temperature
