Advertisements
Advertisements
प्रश्न
Identify the elements with the following property and arrange them in increasing order of their reactivity
- An element which is a soft and reactive metal
- The metal which is an important constituent of limestone
- The metal which exists in liquid state at room temperature
उत्तर
- An element which is a soft and reactive metal is sodium.
- The metal which is an important constituent of limestone is calcium.
- The metal which exists in the liquid state at room temperature is mercury.
The arrangement of these elements in increasing order of their reactivity would be mercury (Hg) < calcium (Ca) < sodium (Na).
APPEARS IN
संबंधित प्रश्न
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
Nature of oxides of the elements ?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.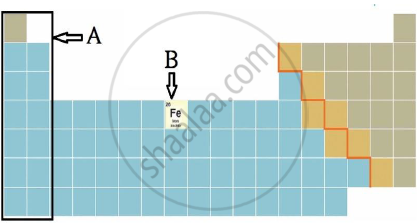
Select the correct answer
Which of the following electronic structure is of a metal?
Match the atomic number with the following:
A metal of valency one.
The tendency of an element to form cation is the _______ character of that element.
The metallic character of the element decreases moving across a period from left to the right.
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing metallic character)
Metals are good ______ because they are electron ______.
Arrange:
Be, Li, C, B, N, O, F (in increasing metallic character).
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last).
Boron, Aluminium, Gallium, Indium, Thallium
Answer the following question in relation to the above group of elements:
Will the elements in the group to the right of this boron group be more metallic or less metallic in character? Justify your answer.
