Advertisements
Advertisements
प्रश्न
Match the atomic number with the following:
A metal of valency one.
विकल्प
19
15
8
4
2
उत्तर
19
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instructions given in the brackets:
Cl, F, Br, I (increasing order of electron affinity)
Which has the maximum metallic character Na, Li or K?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.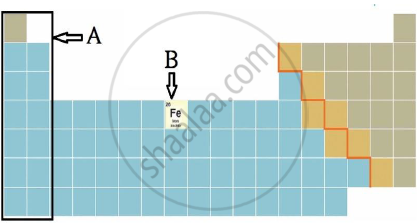
In Period 3, the most metallic element is ______.
Among the elements of the second period, Li to Ne, pick out the element that is the most reactive non-metal
Explain
Larger the atomic size, more metallic is the element.
If an element is in group 7 is it likely to be metallic or non metallic in character?
Write the name.
The atom having the smallest atomic radius from zero group.
Write scientific reason.
The non-metallic character increases while going from left to right in a period.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last).
Boron, Aluminium, Gallium, Indium, Thallium
Answer the following question in relation to the above group of elements:
Will the elements in the group to the right of this boron group be more metallic or less metallic in character? Justify your answer.
