Advertisements
Advertisements
प्रश्न
Write the name.
The atom having the smallest atomic radius from zero group.
उत्तर
The atom having the smallest atomic radius from zero group- Helium (He)
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instructions given in the brackets:
Cl, F, Br, I (increasing order of electron affinity)
Metallic character and non-metallic character are periodic properties discuss ?
Name the periodic property which relates to the character of element which loses one or more electrons when supplied with energy.
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
How does the chemical reactivity of
alkali metals vary?
The elements at the bottom of a group would be expected to show ______ metallic character than the element at the top.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 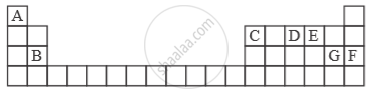
Which non-metallic element has the valency of 2?
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most non-metallic element be found at A,B,C or D ?
In the third period, which is the most metallic and most non-metallic element?
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The element at the bottom of a group would be expected to show ______ metallic character than the element at the top.
Use the information given in (a) to (h) to identify the substances P to W selecting your answers from the given list.
List:
| Calcium | Oxygen | Copper (II) Oxide |
| Carbon | Calcium hydroxide | Copper (II) Nitrate |
| Lead (II) Oxide | Hydrogen chloride | Chlorine |
| Lead (II) Nitrate | Calcium Oxide | Ammonium chloride |
- P is white solid. When heated produces white fumes (sublime).
- P and R on warming produce an alkaline gas.
- On adding water to T, heat is evolved and R is formed.
- Q burns brightly in the air to form T.
- When S is heated, it gives off brown fumes and leaves a black residue of U.
- A solution of S is formed by warming U with dilute nitric acid.
- V is a gaseous non-metallic element that reacts with hydrogen to form W.
- A solution of W will neutralize the solution of R.
The tendency of an element to form cation is the _______ character of that element.
Write the name.
Two elements having two orbits.
Silicon is a metallic element.
Write an Explanation.
Electronegativity
3, 1, 2 electrons are in valence shells of X, Y, Z elements. From this information, state the group in which they belong and write their valencies.
Which of the following set of elements is written in order of their increasing metallic character?
The lightest liquid metal is ______
