Advertisements
Advertisements
प्रश्न
Write the name.
Two elements having two orbits.
उत्तर
Two elements having two orbits- Carbon (C), oxygen (O)
APPEARS IN
संबंधित प्रश्न
Choose the word or phrase from the brackets which correctly complete the following statement:
Metals are good ______ (oxidizing agents/reducing agents) because they are electron ______ (acceptors/donors).
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Answer the following in respect of element `31/15 P `
Is it a metal or non - metal
Name any five periods properties.
Name the periodic property which relates to the character of element which loses one or more electrons when supplied with energy.
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
Which has the maximum metallic character Na, Li or K?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.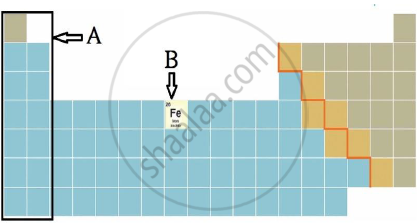
In Period 3, the most metallic element is ______.
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most non-metallic element be found at A,B,C or D ?
Explain
Larger the atomic size, more metallic is the element.
If an element is in group 7 is it likely to be metallic or non metallic in character?
Within a group, where would you expect to find the element with the greatest metallic character.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last). Answer the following question in relation to the given group of elements:
Which element has the most metallic character?
Use the information given in (a) to (h) to identify the substances P to W selecting your answers from the given list.
List:
| Calcium | Oxygen | Copper (II) Oxide |
| Carbon | Calcium hydroxide | Copper (II) Nitrate |
| Lead (II) Oxide | Hydrogen chloride | Chlorine |
| Lead (II) Nitrate | Calcium Oxide | Ammonium chloride |
- P is white solid. When heated produces white fumes (sublime).
- P and R on warming produce an alkaline gas.
- On adding water to T, heat is evolved and R is formed.
- Q burns brightly in the air to form T.
- When S is heated, it gives off brown fumes and leaves a black residue of U.
- A solution of S is formed by warming U with dilute nitric acid.
- V is a gaseous non-metallic element that reacts with hydrogen to form W.
- A solution of W will neutralize the solution of R.
The metallic character of the element decreases moving across a period from left to the right.
A metal ‘M’ is in the first group of the Periodic Table. What will be the formula of its oxide?
Which of the following set of elements is written in order of their increasing metallic character?
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing metallic character)
