Advertisements
Advertisements
प्रश्न
Assertion: Critical temperature of CO2 is 304 K, it can be liquefied above 304 K.
Reason: For a given mass of gas, volume is to directly proportional to pressure at constant temperature
विकल्प
both assertion and reason are true and reason is the correct explanation of assertion
both assertion and reason are true but reason is not the correct explanation of assertion
assertion is true but reason is false
both assertion and reason are false
उत्तर
both assertion and reason are false
APPEARS IN
संबंधित प्रश्न
Convert the following pressure value into Pascals.
10 atmosphere
Convert the following pressure value into Pascals.
1 kPa
Hot air balloons float in the air because of the low density of the air inside the balloon. Explain this with the help of an appropriate gas law.

Identify the gas laws from the following diagram.
| Diagram | Gas laws |
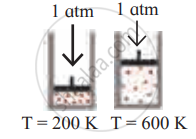 |
______________ |
Consider a sample of a gas in a cylinder with a movable piston.

Show diagrammatically the changes in the position of the piston, if the temperature is decreased from 300 K to 150 K at constant pressure.
State Boyle's law.
Explain the following observation.
The size of a weather balloon becomes larger and larger as it ascends up to larger altitude
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
A certain sample of gas has a volume of 0.2 L at one atmosphere pressure and 273.15 K. What is the volume of gas at 273.15°C at same pressure?
A gas occupies a volume of 4.2 dm3 at 101 kPa pressure. What volume will gas occupy if the pressure is increased to 235 kPa keeping the temperature constant?
