Advertisements
Advertisements
प्रश्न
Convert the following pressure value into Pascals.
10 atmosphere
उत्तर
1 atm = 1.01325 x 105 Pa
∴ 10 atm = 1.01325 x 105 x 10 Pa
= 1.01325 × 106 Pa
10 atmosphere = 1.01325 × 106 Pa
APPEARS IN
संबंधित प्रश्न
Convert the following pressure value into Pascals.
1 kPa
Convert 101.325 kPa to bar.
Hot air balloons float in the air because of the low density of the air inside the balloon. Explain this with the help of an appropriate gas law.

Identify the gas laws from the following diagram.
| Diagram | Gas laws |
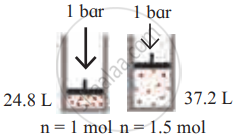 |
______________ |
Write the statement for Charles’ law
With the help of the graph answer the following -

At constant temperature, Identify the law.
Solve the following.
A balloon is inflated with helium gas at room temperature of 25°C and at 1 bar pressure when its initial volume is 2.27L and allowed to rise in the air. As it rises in the air external pressure decreases and the volume of the gas increases till finally, it bursts when external pressure is 0.3bar. What is the limit at which the volume of the balloon can stay inflated?
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
State Boyle's law.
Explain the following observation.
Aerated water bottles are kept under water during summer
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
A small bubble rises from the bottom of a lake where the temperature and pressure are 6°C and 4 atm. to the water surface, where the temperature is 25°C and pressure is 1 atm. Calculate the final volume in (mL) of the bubble, if its initial volume is 1.5 mL.
Hydrochloric acid is treated with a metal to produce hydrogen gas. Suppose a student carries out this reaction and collects a volume of 154.4 × 10−3 dm3 of a gas at a pressure of 742 mm of Hg at a temperature of 298 K. What mass of hydrogen gas (in mg) did the student collect?
For a given mass of an ideal gas, which of the following statements is CORRECT?
A certain sample of gas has a volume of 0.2 L at one atmosphere pressure and 273.15 K. What is the volume of gas at 273.15°C at same pressure?
Volume of a balloon at 25°C and 1 bar pressure is 2.27 L. If the pressure of the gas in balloon is reduced to 0.227 bar, what is the rise in volume of a gas?
A certain mass of a gas occupies a volume of 2 dm3 at STP. At what temperature the volume of gas becomes double, keeping the pressure constant?
Isochor is the graph plotted between ______.
A gas occupies a volume of 4.2 dm3 at 101 kPa pressure. What volume will gas occupy if the pressure is increased to 235 kPa keeping the temperature constant?
The volume of 400 cm3 chlorine gas at 400 mm of Hg is decreased to 200 cm3 at constant temperature. What is the new pressure of gas?
10 g of gas at one atomospheric pressure is cooled from 273.15°C to 0°C keeping the volume constant. What is the final pressure?
At what temperature, the volume of gas would become zero?
The number of molecules in 8.96 litres of gas at 0°C and 1 atm. pressure is approximately ______.
