Advertisements
Advertisements
प्रश्न
Assertion: Network polymers are thermosetting.
Reason: Network polymers have high molecular mass.
विकल्प
Assertion and reason both are correct statement but reason does not explain assertion.
Assertion and reason both are correct statements and reason explains the assertion.
Both assertion and reason are wrong statement.
Assertion is correct statement and reason is wrong statement.
Assertion is wrong statement and reason is correct statement.
उत्तर
Assertion and reason both are correct statement but reason does not explain assertion.
Explanation:
Extensive cross linking during polymerisation leads to the formation of three-dimensional network which is hard, infusible and insoluble.
APPEARS IN
संबंधित प्रश्न
Write the names and structures of the monomers of the following polymers: Polyvinyl chloride
Write the names of monomers used for getting the following polymers:
Teflon
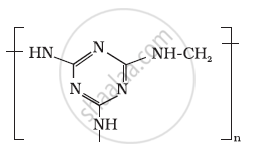
Identify the monomer in the following polymeric structures.
Write a chemical reaction to show that the open structure of D-glucose contains the following :
Straight chain
A natural linear polymer of 2-methyl-1, 3-butadiene becomes hard on treatment with sulphur between 373 to 415 K and – S – S – bonds are formed between chains. Write the structure of the product of this treatment?
Identify the type of polymer given in the following figure.

What is the structural difference between HDP and LDP? How does the structure account for different behaviour and nature, hence the use of a polymer?
Which of the following is a cross linked polymer?
Which one is disperse dye?
Linear chain polymers have ______.
