Advertisements
Advertisements
प्रश्न
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
विकल्प
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Both assertion and reason are correct statements but reason is not correct explanation of assertion.
उत्तर
Assertion and reason both are correct and reason is correct explanation of assertion.
Explanation:
P-nitro phenol is more acidic as nitro group helps in the stabilization of the phenoxide ion by dispersal of negative charge due to resonance.
APPEARS IN
संबंधित प्रश्न
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\phantom{............}\ce{CH2OH}\\
\phantom{......}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{......}|\phantom{............}|\phantom{.}\\
\phantom{........}\ce{CH2Cl}\phantom{......}\ce{CH3}\phantom{}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{......}|\phantom{..}\\
\ce{OH}\phantom{...}\ce{OH}\phantom{}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{...}|\\
\phantom{.....}\ce{CH3}
\end{array}\]
Write the IUPAC name of the following compound:
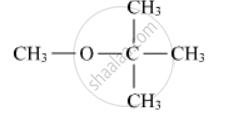
Butane-2-ol is ____________.
1-Propanol and 2-propanol can be best distinguished by:
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
Write structural formulae for:
p-Nitrophenol
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
