Advertisements
Advertisements
प्रश्न
Balance the following chemical equation:

उत्तर
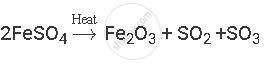
APPEARS IN
संबंधित प्रश्न
Balance the given equation:
AI(OH)3  AI2O3 +H2O
AI2O3 +H2O
Name the various types of chemical reactions.
Balance the following chemical equation :
Na2CO3 + HCl → NaCI + H2O + CO2
Write the balanced chemical equation of the following reaction.
iron pyrites(FeS2) + oxygen → ferric oxide + sulphur dioxide
Write the balanced chemical equation of the following reaction.
potassium dichromate + hydrochloric acid → Potassium chloride + chromium chloride + water + chlorine
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Lead nitrate to ammonium hydroxide.?
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Heat energy
Write a balanced equation for the following word equation:
Potassium bicarbonate → Potassium carbonate + Water + Carbon dioxide
Complete the given chemical reaction.
\[\ce{CuSO_{4(aq)} + Fe_{(s)} ->}\]______ + ______
Three beakers labelled as A, B and C each containing 25 mL of water were taken. A small amount of NaOH, anhydrous CuSO4 and NaCl were added to the beakers A, B and C respectively. It was observed that there was an increase in the temperature of the solutions contained in beakers A and B, whereas in case of beaker C, the temperature of the solution falls. Which one of the following statement(s) is(are) correct?
- In beakers A and B, exothermic process has occurred.
- In beakers A and B, endothermic process has occurred.
- In beaker C exothermic process has occurred.
- In beaker C endothermic process has occurred.
