Advertisements
Advertisements
प्रश्न
Balance the following chemical equation:

उत्तर
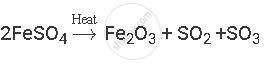
APPEARS IN
संबंधित प्रश्न
What is wrong with the following chemical equation?
Mg + O → MgO
Correct and balance it.
Is Burning of natural gas an endothermic reaction or an exothermic reaction?
The chemical reaction between quicklime and water is characterised by:
(a) evolution of hydrogen gas
(b) formation of slaked lime precipitate
(c) change in temperature of mixture
(d) change in colour of the product
When metal X is treated with a dilute acid Y, then a gas Z is evolved which burns readily by making a little explosion.
(a) Name any two metals which can behave like metal X.
(b) Name any two acids which can behave like acid Y.
(c) Name the gas Z.
(d) Is the gas Z lighter than or heavier than air?
(e) Is the reaction between metal X and dilute acid Y, exothermic or endothermic?
(f) By taking a specific example of metal X and dilute acid Y, write a balanced chemical equation for the reaction which takes place. Also indicate physical states of all the reactants and products.
Write word equation for the following skeletal equation:
\[\ce{Zn + HCl -> ZnCl2 + H2}\]
In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – mercury [II] oxide.
\[\ce{2KClO3->[MnO2] 2KCl + 3O2[g] - is a balanced equation.}\]
State what is a ‘balanced equation’.
Write a balanced equation for the following word equation:
Iron + Hydrochloric acid → Iron [II] chloride + Hydrogen
Write an Explanation.
Balanced equations
