Advertisements
Advertisements
प्रश्न
In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
उत्तर
The balanced chemical equation for the electrolysis of water is: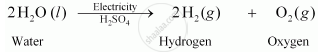
It can be observed from the balanced chemical equation that the volumes of hydrogen and oxygen produced during electrolysis are in the ratio 2: 1. Thus, the volume of hydrogen gas collected in the process is double than that of oxygen.
APPEARS IN
संबंधित प्रश्न
What is a balanced chemical equation? Why should a chemical equation be balanced?
How will you indicate a solution made in water in a chemical equation?
Translate the following statement into chemical equation and then balance the equation:
Carbon disulphide burns in air to give carbon dioxide and sulphur dioxide.
Complete and balance the following equation:
NaOH + ............  Na2 SO4 + H2O
Na2 SO4 + H2O
Balance the following equation and add state symbols:
Zn + HCI → ZnCI2 + H2
Is Respiration an endothermic reaction or an exothermic reaction?
An acid which can decolourise purple coloured potassium permanganate solution is:
(a) sulphuric acid
(b) citric acid
(c) carbonic acid
(d) hydrochloric acid
Balance the following equation:
Fe + H2O → Fe3O4 + H2
Balance the following equation:
PbS + O2 → PbO + SO2
Balance the following simple equation:
Mg + N2 → Mg3N2
