Advertisements
Advertisements
Question
In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
Solution
The balanced chemical equation for the electrolysis of water is: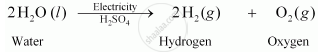
It can be observed from the balanced chemical equation that the volumes of hydrogen and oxygen produced during electrolysis are in the ratio 2: 1. Thus, the volume of hydrogen gas collected in the process is double than that of oxygen.
APPEARS IN
RELATED QUESTIONS
How will you indicate a solution made in water in a chemical equation?
How will you indicate Endothermic reaction in a chemical equation?
Give one example of a chemical reaction characterised by change in state.
Write symbolic representation for the following word equation and balance them :
Calcium oxide + Water → Calcium hydroxide
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Write the balanced chemical equation of the following reaction.
potassium dichromate + sulphuric acid → potassium sulphate + chromium sulphate + water + oxygen.
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Iron [III] chloride to ammonium hydroxide.
Name the following:
The gas evolved when a dilute acid is added to chalk [limestone].
Balance the following simple equation:
Al + O2 → Al2O3
Give a balanced equation by the partial equation method, [steps are given below].
Oxidation of Lead [II] Sulphide by Ozone
- Ozone first decomposes to give molecular oxygen & nascent oxygen.
- Nascent oxygen then oxidises lead [II] sulphide to lead [II] sulphate.
