Advertisements
Advertisements
प्रश्न
In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
उत्तर
The balanced chemical equation for the electrolysis of water is: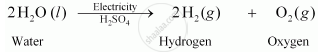
It can be observed from the balanced chemical equation that the volumes of hydrogen and oxygen produced during electrolysis are in the ratio 2: 1. Thus, the volume of hydrogen gas collected in the process is double than that of oxygen.
APPEARS IN
संबंधित प्रश्न
Balance the following chemical equation.
\[\ce{BaCl2 + H2SO4 -> BaSO4 + HCl}\]
How will you indicate Exothermic reaction in a chemical equation?
Give one example of a chemical reaction.
State one characteristic of the chemical reaction which takes place when lemon juice is added gradually to potassium permanganate solution.
State one characteristic of the chemical reaction which takes place when dilute sulphuric acid is added to barium chloride solution.
Give one example of an exothermic reaction.
Write word equation for the following skeletal equation:
\[\ce{KNO3 -> KNO2 + O2}\]
Balance the following equation:
KOH + Cl2 → KCl + KClO + H2O
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
Writing a chemical reaction in brief by using chemical formulae is called as _______.
