Advertisements
Advertisements
प्रश्न
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
उत्तर
MnO2 = Mn + 2(O)
= 55 + 2(16)
= 55 + 32
= 87 g
1 mole of MnO2 weighs = 87 g (mol. Mass)
So, 0.02 mole will weigh = 87 × 0.02 = 1.74 g
APPEARS IN
संबंधित प्रश्न
Write complete balanced equation for the following reaction:
Sulphur dioxide (gas) + Oxgyen (gas)  Sulphur trioxide (gas)
Sulphur trioxide (gas)
Balance the given equation:
HNO3 + Ca(OH)2  Ca(NO3)2 + H2O
Ca(NO3)2 + H2O
The metal M reacts vigorously with water to form a solution S and a gas G. The solution S turns red litmus to blue whereas gas G, which is lighter than air, burns with a pop sound. Metal M has a low melting point and it is used as a coolant in nuclear reactors.
(a) What is metal M?
(b) What is solution S? Is it acidic or alkaline?
(c) What is gas G?
(d) Write a balanced chemical equation for the reaction which takes place when metal M reacts with water.
(e) Is this reaction exothermic or endothermic?
Write balanced chemical equation with state symbols for the following reaction:
Barium chloride solution reacts with sodium sulphate solution to give insoluble barium sulphate and a solution of sodium chloride.
Write word equation for the following skeletal equation:
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Balance the following equation. Also name the product formed.
`"KCIO"_3 → "KCI" +"O"_2`
Write your observation and name the product when zinc reacts with dilute hydrochloric acid.
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
2H2O + 2Cl2 → 2HCl + O2
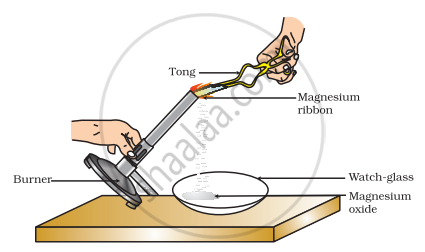
Which of the following is the correct observation of the reaction shown in the above set up?
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Grams: 42 g + ______ `→` ______ + ______
