Advertisements
Advertisements
प्रश्न
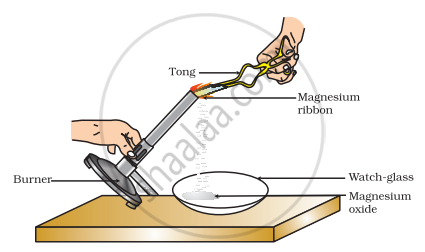
Which of the following is the correct observation of the reaction shown in the above set up?
विकल्प
Brown powder of Magnesium oxide is formed.
Colourless gas which turns lime water milky is evolved.
Magnesium ribbon burns with brilliant white light.
Reddish brown gas with a smell of burning Sulphur has evolved.
उत्तर
Magnesium ribbon burns with brilliant white light.
APPEARS IN
संबंधित प्रश्न
Balance the given equation:
Mg(OH)2 + HCI MgCI + H2O
Aluminium burns in chlorine to form aluminium chloride (AlCl3). Write a balanced chemical equation for this reaction.
Convey the following information in the form of a balanced chemical equation:
"An aqueous solution of ferrous sulphate reacts with an aqueous solution of sodium hydroxide to form a precipitate of ferrous hydroxide and sodium sulphate remains in solution."
Write your observations and name the product when
Iron nails are added to an aqueous solution of copper sulphate.
Balance the following equation. Also name the product formed.
`"CO" + "O"_2 → " CO"_2`
Balance the following equation:
PbS + O2 → PbO + SO2
Explain the term ‘chemical equation’.
Balance the following simple equation:
Al + H2O → Al2O3 + H2
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write the number of atoms of various elements in the reactants and products.
What is a product in a chemical equation?
