Advertisements
Advertisements
Question
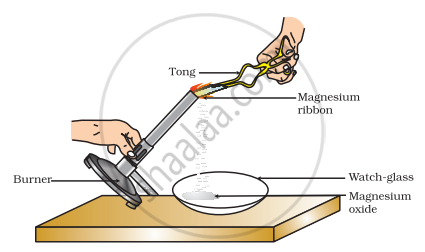
Which of the following is the correct observation of the reaction shown in the above set up?
Options
Brown powder of Magnesium oxide is formed.
Colourless gas which turns lime water milky is evolved.
Magnesium ribbon burns with brilliant white light.
Reddish brown gas with a smell of burning Sulphur has evolved.
Solution
Magnesium ribbon burns with brilliant white light.
APPEARS IN
RELATED QUESTIONS
Methyl orange is used as an indicator. It shows colour change in acid, base and neutral substance. Tabulate your result as follows:
| Indicator | Colour change | Inference |
| Methyl Orange | No change | ...................... |
| .................... | Acid | |
| Yellow | ....................... |
Balance the given equation:
AI2(SO4)3 +NaOH  AI(OH)3 + Na2SO4
AI(OH)3 + Na2SO4
Balance the given equation:
BaCI2 + H2SO4  BaSO4 + HCI
BaSO4 + HCI
Write the balanced chemical equation of the following reaction.
potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
Write the balanced chemical equation of the following reaction.
aluminium carbide + water → aluminium hydroxide + methane
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of acid required.
(a) Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen Chloride
- Chlorine
- Carbon Dioxide
- Oxygen
- Hydrogen
(b) Select a basic gas mentioned in Q.1 (a). How is the basic nature suspected?
(c)Select acidic gases from the gases mentioned in Q.1 (a). How is the acidic nature suspected?
(d) State the gas responsible for bleaching action.
(e)Which gas turn blue cobalt chloride paper light pink?
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Iron [III] chloride to ammonium hydroxide.
Name the following:
A catalyst which increases the rate of a chemical reaction.
Balance the following simple equation:
Fe + H2O ⇌ Fe3O4 + H2
