Advertisements
Advertisements
प्रश्न
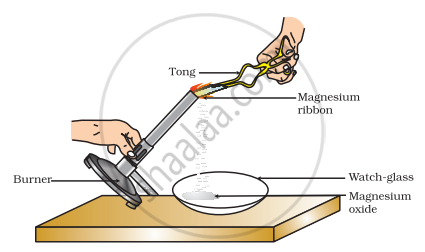
Which of the following is the correct observation of the reaction shown in the above set up?
पर्याय
Brown powder of Magnesium oxide is formed.
Colourless gas which turns lime water milky is evolved.
Magnesium ribbon burns with brilliant white light.
Reddish brown gas with a smell of burning Sulphur has evolved.
उत्तर
Magnesium ribbon burns with brilliant white light.
APPEARS IN
संबंधित प्रश्न
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
Dry raisins were soaked in water for 2 hours, to determine the percentage of water absorbed by raisins. Before final weighing of swollen raisins, the extra water left on the surface of soaked raisins was removed by:
A student weighed some raisins and recorded the weight as ‘x’. She then soaked the raisins in distilled water. After about 2 hours she removed the raisins, wiped them dry and weighed them again, and recorded that as ‘y’. The percentage of water absorbed by raisins may be determined using the relationship.
Balance the equation stepwise.
SO2(g) + H2S(aq) → S(s) + H2O(l)
Choose the correct answer from the options given below.
The formula of sodium carbonate is Na2CO3 and that of calcium hydrogen carbonate is
Write the balanced chemical equation of the following reaction.
aluminium sulphate + sodium hydroxide → sodium sulphate + sodium meta aluminate + water.
Write the balanced chemical equation of the following reaction.
aluminium + sodium hydroxide + water → sodium meta aluminate + hydrogen
Give a balanced equation for addition of iron to copper [II] sulphate solution. State the change in colour seen.
Name the following:
Two gases which react under pressure in presence of a catalyst at elevated temperatures to give a gaseous product.
Balance the following simple equation:
FeCl2 + Cl2 → FeCl3
