Advertisements
Advertisements
प्रश्न
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
उत्तर
A chemical equation can be made more informative by:
- Indicating physical states of reactants and products.
- Indicating heat change in the reaction.
- Indicating the reaction condition.
Example:
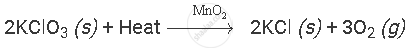
where,
(s) and (g) signifies solid and gaseous state of compounds, respectively.
Heat written on reactants' side signifies that the reaction consumes heat.
MnO2 written above right-handed arrow signifies reaction takes place in the presence of MnO2.
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance it.
Hydrogen sulphide gas burns in the air to give water and sulphur dioxide.
To determine the percentage of water absorbed by raisins, raisins are soaked in water for:
Balance the following equation:
Fe2O3 + CO → Fe + CO2
Balance the following equation:
P + HNO3 → NO2 + H2O + H3PO4
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
\[\ce{C +2S -> CS2}\]
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
\[\ce{2SO2 + O2 ⇌[V2O5][450°C] 2SO3}\]
What is meant by ‘reactants’ and ‘products’ in a chemical equation?
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
K + H2O → KOH + H2
The following reaction is an example of a `4"NH"_3("g") + "SO"_2 -> 4"NO"("g") + 6"H"_2"O"("g")`
- displacement reaction
- combination reaction
- redox reaction
- neutralisation reaction
