Advertisements
Advertisements
प्रश्न
What are the various ways in which a chemical equation can be made more informative? Give examples to illustrate your answer.
उत्तर
A chemical equation can be made more informative by:
- Indicating physical states of reactants and products.
- Indicating heat change in the reaction.
- Indicating the reaction condition.
Example:
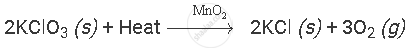
where,
(s) and (g) signifies solid and gaseous state of compounds, respectively.
Heat written on reactants' side signifies that the reaction consumes heat.
MnO2 written above right-handed arrow signifies reaction takes place in the presence of MnO2.
APPEARS IN
संबंधित प्रश्न
Write word equation for the following skeletal equation:
\[\ce{KNO3 -> KNO2 + O2}\]
The step(s) necessary for determining the percentage of water absorbed by raisins is/are:
Balance the following equation:
S + H2SO4 → SO2 + H2O
Write the balanced chemical equation of the following reaction.
sodium chloride + manganese dioxide + sulphuric acid → sodium hydrogen sulphate + manganese sulphate + water + chlorine.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of salt formed.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the mass of chlorine gas formed.
What is thermit process? Where is this process used? Write a balanced chemical equation for the reaction involved.
Name the following:
The silvery residue obtained on heating mercury [II] oxide.
\[\ce{2KClO3->[MnO2] 2KCl + 3O2[g] - is a balanced equation.}\]
State why the compound MnO2 is written above the arrow.
Balance the following simple equation:
N2 + O2 ⇌ NO
