Advertisements
Advertisements
प्रश्न
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate mass of MnO2 used.
उत्तर
MnO2 = Mn + 2(O)
= 55 + 2(16)
= 55 + 32
= 87 g
1 mole of MnO2 weighs = 87 g (mol. Mass)
So, 0.02 mole will weigh = 87 × 0.02 = 1.74 g
APPEARS IN
संबंधित प्रश्न
What does the symbol (aq) represent in a chemical equation?
Balance the given chemical equation:
NH3 → N2 + H2
Give one example of a chemical reaction characterised by formation of a precipitate.
The chemical reaction between quicklime and water is characterised by:
(a) evolution of hydrogen gas
(b) formation of slaked lime precipitate
(c) change in temperature of mixture
(d) change in colour of the product
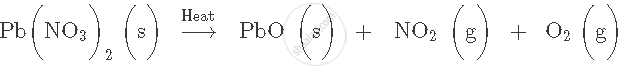
Balance the following chemical equation:
A white precipitate can be obtained by adding dilute sulphuric acid to:
(a) CuSO4 solution
(b) Nacl solution
(c) BaCl2 solution
(d) Na2SO4 solution
A student weighed some raisins and recorded the weight as ‘x’. She then soaked the raisins in distilled water. After about 2 hours she removed the raisins, wiped them dry and weighed them again, and recorded that as ‘y’. The percentage of water absorbed by raisins may be determined using the relationship.
To determine the percentage of water absorbed by raisins, raisins are soaked in water for:
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
Fe2O3 + 2H2 → 2Fe + 3H2O
Which of the following are exothermic processes?
- Reaction of water with quick lime
- Dilution of an acid
- Evaporation of water
- Sublimation of camphor (crystals)
