Advertisements
Advertisements
प्रश्न
Balance the following chemical equation:
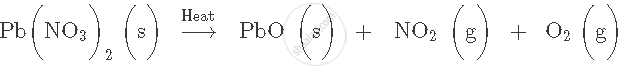
उत्तर
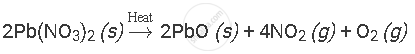
APPEARS IN
संबंधित प्रश्न
Translate the following statement into chemical equation and then balance it.
Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
Write word equation for the following skeletal equation:
\[\ce{KClO3 -> KCl + O2}\]
Balance the following equation:
KOH + Cl2 → KCl + KClO + H2O
Write the balanced chemical equations of the following reactions.
chlorine + sulphur dioxide + water → sulphuric acid + hydrogen chloride
Give an example of a chemical equation in which two reactants form:
four product
Balance the following simple equation:
Al + H2SO4 → Al2(SO4)3 + H2
Balance the following simple equation:
KBr + Cl2 → KCl + Br2
Write a balanced equation for the following word equation:
Iron + Hydrochloric acid → Iron [II] chloride + Hydrogen
Give a balanced equation by partial equation method, [steps are given below].
The reaction of excess ammonia with chlorine – Ammonia as a reducing agent
- Ammonia first reacts with chlorine to give hydrogen chloride and nitrogen.
- Hydrogen chloride then further reacts with excess ammonia to give ammonium chloride.
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write the number of atoms of various elements in the reactants and products.
