Advertisements
Advertisements
Question
Balance the following chemical equation:
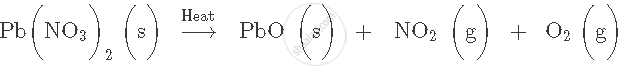
Solution
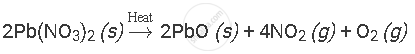
APPEARS IN
RELATED QUESTIONS
Translate the following statement into chemical equation and then balance it.
Hydrogen sulphide gas burns in the air to give water and sulphur dioxide.
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in colour
Give chemical equation for the reaction involved in the above case.
A white precipitate will be formed if we add common salt solution to:
(a) Ba(NO3)2 solution
(b) KNO3 solution
(c) AgNO3 solution
(d) Mg(NO3)2 solution
Write the balanced chemical equation of the following reaction.
potassium dichromate + hydrochloric acid → Potassium chloride + chromium chloride + water + chlorine
The chemical reactions in which heat is liberated are called endothermic reactions.
Write word equation for the following molecular equation:
CaO + H20 → Ca (OH)2 + Δ
Word equation:
State why [+ Δ ] is not written in the reaction, if ZnO reacts with H2O.
Write word equation for the following molecular equation:
\[\ce{CuSO4 + 2NaOH -> Na2SO4 + Cu(OH)2↓}\]
Word equation:
State the colour of the products.
Name the following:
Two gases which react under pressure in presence of a catalyst at elevated temperatures to give a gaseous product.
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
6NaOH + 3Cl2 → 6NaCl + NaClO3 + 3H2O
To balance the following chemical equation the value of x and y should respectively be:
\[\ce{2NaOH + xAl_2O_3->yNaAlO_2 + H_2O}\]
