Advertisements
Advertisements
प्रश्न
How will you indicate a solution made in water in a chemical equation?
उत्तर
A solution made in water is indicated by writing (aq) after its formula in a chemical equation.
Example: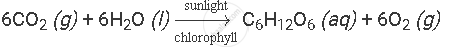
APPEARS IN
संबंधित प्रश्न
Balance the chemical equation.
\[\ce{HNO3 +Ca(OH)2 -> Ca(NO3)2 + H2O}\]
Which of the following is not an endothermic reaction?
(a) CaCO3 → CaO + CO2
(b) 2H2O →2H2 + O2
(c) 6CO2 + 6H2O → C6H12O6 + 6O2
(d) C6H12O6 + 6O2 → 6CO2 + 6H2O
When metal X is treated with a dilute acid Y, then a gas Z is evolved which burns readily by making a little explosion.
(a) Name any two metals which can behave like metal X.
(b) Name any two acids which can behave like acid Y.
(c) Name the gas Z.
(d) Is the gas Z lighter than or heavier than air?
(e) Is the reaction between metal X and dilute acid Y, exothermic or endothermic?
(f) By taking a specific example of metal X and dilute acid Y, write a balanced chemical equation for the reaction which takes place. Also indicate physical states of all the reactants and products.
Write symbolic representation for the following word equation and balance them :
Calcium oxide + Water → Calcium hydroxide
Balance the following equation. Also name the product formed.
`"NaNO"_3 → "NaNO"_2 + "O"_2` Name of product (s)
Balance the following equation:
KOH + Cl2 → KCl + KClO + H2O
Balance the following equation:
HNO3 + H2S → NO2 + H2O + S
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
\[\ce{2H2O ->[electric][current] 2H2 + O2}\]
Give word equation for the following chemical reaction and give the names of the product formed.
Zn + 2HC1→ ZnCl2 + H2
To balance the following chemical equation the value of x and y should respectively be:
\[\ce{2NaOH + xAl_2O_3->yNaAlO_2 + H_2O}\]
