Advertisements
Advertisements
प्रश्न
Calculate the emf of the following cell at 25°C :
उत्तर
For the given cell representation, the cell reaction will be
Zn(s) + 2H+(aq) → Zn2+(aq) + H2(g)
The standard emf of the cell will be
\[E_{cell}^o = E_{H^+ / H_2}^o - E_{{Zn}^{2 +} /Zn}^o \]
\[ E_{cell}^o = 0 - \left( - 0 . 76 \right) = 0 . 76 V\]
The Nernst equation for the cell reaction at 25 ºC will be
\[E_{cell} = E_{cell}^o - \frac{0 . 0591}{n}\log\frac{\left[ {Zn}^{2 +} \right]}{\left[ H^+ \right]^2}\]
\[ = 0 . 76 - \frac{0 . 0591}{2}\log\frac{0 . 001}{\left( 0 . 01 \right)^2} \]
\[ = 0 . 76 - 0 . 02955\left( \log10 \right)\]
\[ = 0 . 76 - 0 . 02955\left( 1 \right)\]
\[ = 0 . 73045 V \approx 0 . 73 V\]
Thus, the emf of the cell at 25 ºC is 0.73 V.
APPEARS IN
संबंधित प्रश्न
Calculate Ecell and ΔG for the following at 28°C :
Mg(s) + Sn2+( 0.04M ) → Mg2+( 0.06M ) + Sn(s)
E°cell = 2.23V. Is the reaction spontaneous ?
Calculate emf of the following cell at 25°C:
\[\ce{Sn/Sn^2+ (0.001 M) || H+ (0.01 M) | H2_{(g)} (1 bar) | Pt_{(s)}}\]
Given: \[\ce{E^\circ(Sn^2+/sn) = -0.14 V, E^\circ H+/H2 = 0.00 V (log 10 = 1)}\]
Calculate emf of the following cell at 25 °C :
Fe|Fe2+(0.001 M)| |H+(0.01 M)|H2(g) (1 bar)|Pt (s)
E°(Fe2+| Fe)= −0.44 V E°(H+ | H2) = 0.00 V
Standard hydrogen electrode operated under standard conditions of 1 atm H2 pressure, 298 K, and pH = 0 has a cell potential of ____________.
Using the data given below find out the strongest reducing agent.
`"E"_("Cr"_2"O"_7^(2-)//"Cr"^(3+))^⊖` = 1.33 V `"E"_("Cl"_2//"Cl"^-) = 1.36` V
`"E"_("MnO"_4^-//"Mn"^(2+))` = 1.51 V `"E"_("Cr"^(3+)//"Cr")` = - 0.74 V
What does the negative sign in the expression `"E"^Θ ("Zn"^(2+))//("Zn")` = − 0.76 V mean?
Which reference electrode is used to measure the electrode potential of other electrodes?
Assertion: Cu is less reactive than hydrogen.
Reason: `E_((Cu^(2+))/(Cu))^Θ` is negative.
Consider the figure and answer the following question.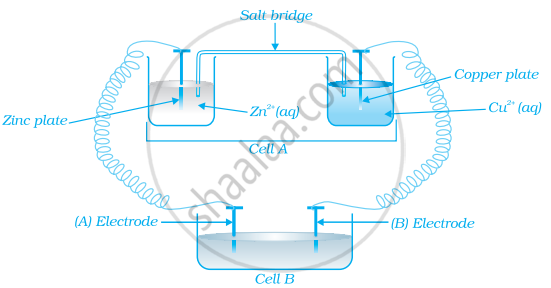
If cell ‘A’ has ECell = 0.5V and cell ‘B’ has ECell = 1.1V then what will be the reactions at anode and cathode?
The emf of a galvanic cell, with electrode potential of Zn2+ = - 0.76 V and that of Cu2+ = 0.34 V, is ______.
