Advertisements
Advertisements
प्रश्न
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
विकल्प
True
False
उत्तर
False
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
What do you understand by atomic size? State its unit?
Which is greater in size Fe2+ or Fe3+?
Arrange the following in order of increasing radii:
CI- , CI
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
The size of an atom depends on the number of valence electrons.
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
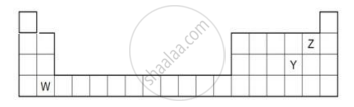
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?