Advertisements
Advertisements
प्रश्न
What do you understand by atomic size? State its unit?
उत्तर
Atomic size is the distance between the centre of an atom's nucleus and its outermost shell. It is measured in Angstroms and Picometres.
संबंधित प्रश्न
Give reason for the following:
Argon atom is bigger than chlorine atom.
Arrange the following in order of increasing radii:
CI- , CI
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
_______ is the distance between the nucleus of the atom and its outermost shell.
Write scientific reason.
In same period, boron and oxygen elements have different atomic size.
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
Which of the following are the characteristics of isotopes of an element?
- Isotopes of an element have same atomic masses
- Isotopes of an element have same atomic number
- Isotopes of an element show same physical properties
- Isotopes of an element show same chemical properties
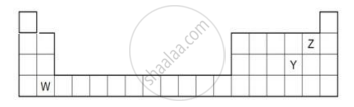
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Which one has the largest size?
