Advertisements
Advertisements
प्रश्न
Give reason for the following:
Argon atom is bigger than chlorine atom.
उत्तर
Since Argon has stable octet configuration, so due to the inter- electronic repulsions the effect of nuclear pull over the valence shell electrons cannot be seen which results in the bigger size.
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Arrange the elements of second and third periods in increasing order of their atomic size (excluding noble gases).
Give scientific reasons for the following:
Atomic radius goes on decreasing while going from left to right in a period.
What happens to the atomic size of elements on moving from left to right in a period?
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
Atomic radius is expressed in the unit _______.
Moving from left to right, the size of the atom decreases.
Write an Explanation.
Atomic radius
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.

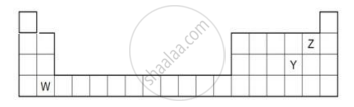
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?