Advertisements
Advertisements
प्रश्न
Give reason for the following:
Argon atom is bigger than chlorine atom.
उत्तर
Since Argon has stable octet configuration, so due to the inter- electronic repulsions the effect of nuclear pull over the valence shell electrons cannot be seen which results in the bigger size.
संबंधित प्रश्न
Arrange the following as per the instruction given in the brackets:
Na, K, Li (Increasing atomic size)
Which is greater in size an atom or a cation?
The size of an atom is indicated by its _______.
While going from top to bottom in a group the atomic radius _______.
Moving from left to right, the size of the atom decreases.
Write an Explanation.
Atomic radius
- In below ladder symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their atomic number in the Periodic Table.
- Arrange them in the order of their group also.

- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
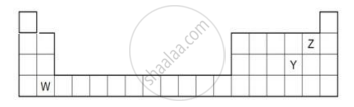
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Give reason for the following:
The size of a Cl− ion is greater than the size of a Cl atom.
