Advertisements
Advertisements
प्रश्न
Moving from left to right, the size of the atom decreases.
विकल्प
Right
Wrong
उत्तर
Moving from left to right, the size of the atom decreases- Right
APPEARS IN
संबंधित प्रश्न
Give the trends in atomic size on moving across the period left to right.
Which is greater in size an atom or an anion?
Which is greater in size Fe2+ or Fe3+?
Fill in the blank:
On moving across a period from right to left in the periodic table, the atomic size of the atom ___________.
Give reasons for the following:
The size of the anion is greater than the size of the parent atom.
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The atom having the smallest size.
Write the name and symbol of the element from the description.
The noble gas with the smallest atomic radius.
Write scientific reason.
Atomic radius goes on increasing down a group.
Among the elements of the second period, Li to Ne, pick out the element with the largest atomic size
Fill in the blank by selecting the correct word from the bracket.
If an element has seven electrons in its outermost shell then it is likely to have the _____ atomic size among all the elements in the same period.
The size of an atom is indicated by its _______.
_______ is the distance between the nucleus of the atom and its outermost shell.
Write an Explanation.
Atomic radius
Which of the following is the correct order of atomic size?
Which of the following gives the correct increasing order of the atomic radii of O, F, and N?
Which among the following elements has the largest atomic radii?
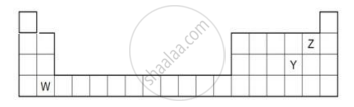
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?
